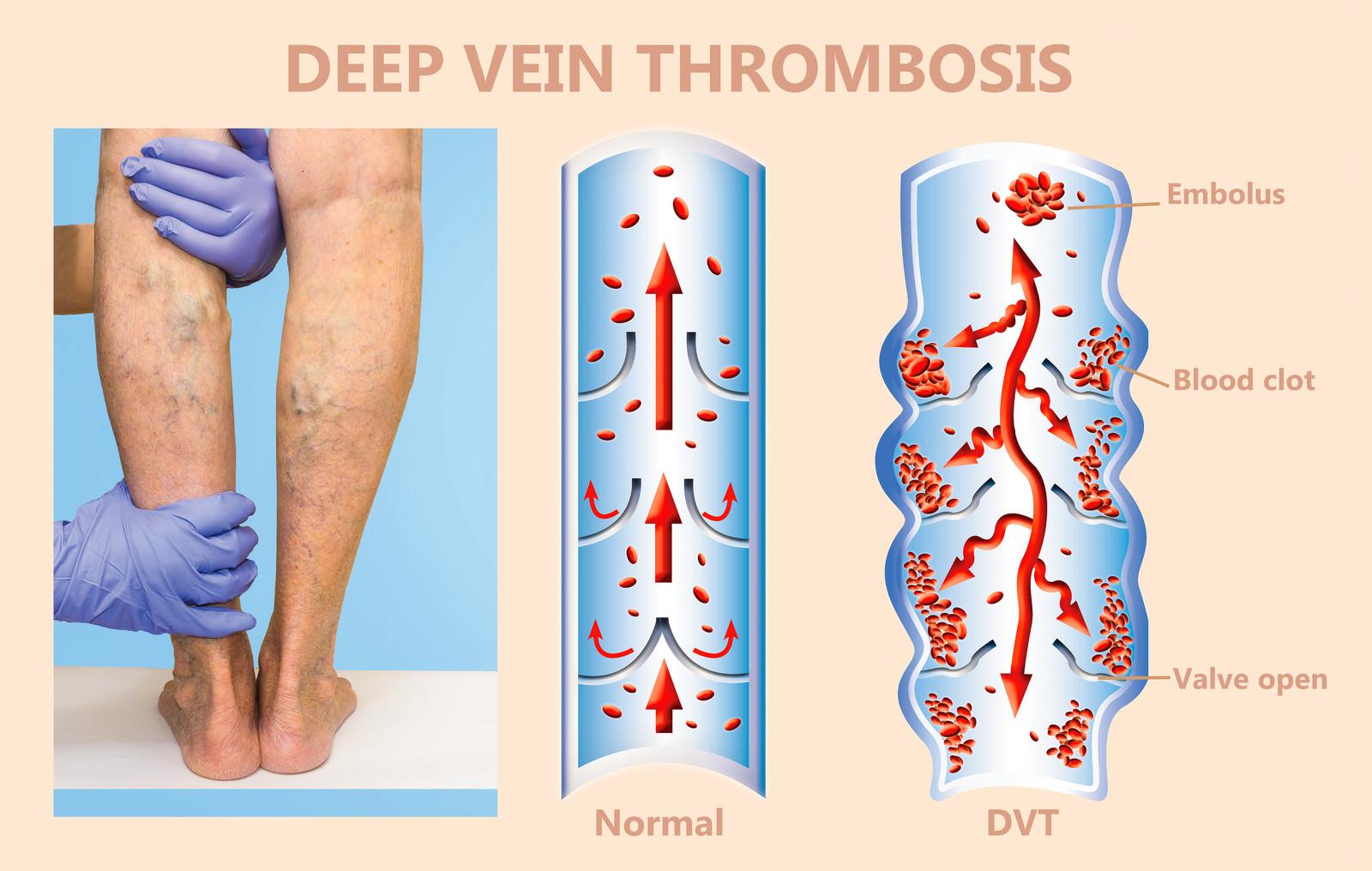
The PRAC noted that the blood clots occurred in veins in the brain (cerebral venous sinus thrombosis, CVST) and the abdomen (splanchnic vein thrombosis) and in arteries, together with low levels of blood platelets and sometimes bleeding. People who have received the vaccine should seek medical assistance immediately if they develop symptoms of this combination of blood clots and low blood platelets (see below). Based on the currently available evidence, specific risk factors have not been confirmed. So far, most of the cases reported have occurred in women under 60 years of age within 2 weeks of vaccination. In reaching its conclusion, the committee took into consideration all currently available evidence, including the advice from an ad hoc expert group.ĮMA is reminding healthcare professionals and people receiving the vaccine to remain aware of the possibility of very rare cases of blood clots combined with low levels of blood platelets occurring within 2 weeks of vaccination. Nothing in this guideline should be interpreted in a way that would be inconsistent with complying with those duties.Ĭommissioners and providers have a responsibility to promote an environmentally sustainable health and care system and should assess and reduce the environmental impact of implementing NICE recommendations wherever possible.EMA confirms overall benefit-risk remains positiveĮMA’s safety committee ( PRAC) has concluded today that unusual blood clots with low blood platelets should be listed as very rare side effects of Vaxzevria (formerly COVID-19 Vaccine AstraZeneca). They should do so in the context of local and national priorities for funding and developing services, and in light of their duties to have due regard to the need to eliminate unlawful discrimination, to advance equality of opportunity and to reduce health inequalities. Local commissioners and providers of healthcare have a responsibility to enable the guideline to be applied when individual professionals and people using services wish to use it. It is not mandatory to apply the recommendations, and the guideline does not override the responsibility to make decisions appropriate to the circumstances of the individual, in consultation with them and their families and carers or guardian.Īll problems (adverse events) related to a medicine or medical device used for treatment or in a procedure should be reported to the Medicines and Healthcare products Regulatory Agency using the Yellow Card Scheme. When exercising their judgement, professionals and practitioners are expected to take this guideline fully into account, alongside the individual needs, preferences and values of their patients or the people using their service. The recommendations in this guideline represent the view of NICE, arrived at after careful consideration of the evidence available. thrombolytic therapy, inferior vena caval filters, investigations for cancer and thrombophilia testing.information and support for people having anticoagulation treatment.long-term anticoagulation for secondary prevention.anticoagulation treatment for suspected or confirmed DVT or PE.diagnosing and managing suspected DVT and PE.This guideline includes recommendations on: See the MHRA advice on warfarin and other anticoagulants – monitoring of patients during the COVID-19 pandemic, which includes reports of supratherapeutic anticoagulation with warfarin. For more details, see the update information. We also clarified the recommendation on the use of the pulmonary embolism rule-out criteria (PERC). In August 2023, we updated recommendations on the use of Wells score and D-dimer in the diagnostic pathways for pulmonary embolism and deep vein thrombosis, following a review of the evidence for people with COVID-19. The guideline does not cover pregnant women. It also covers testing for conditions that can make a DVT or PE more likely, such as thrombophilia (a blood clotting disorder) and cancer. It aims to support rapid diagnosis and effective treatment for people who develop deep vein thrombosis (DVT) or pulmonary embolism (PE). This guideline covers diagnosing and managing venous thromboembolic diseases in adults.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
